Hello, Green Hydrogen
November 30, 2021
This is a game-changer.
New forms of technology in the energy transition have been innovated at a rapid pace. At the forefront of the innovation is a form of energy storage: green hydrogen. This isn’t the first time hydrogen gas has been used in the energy field. Previous instances have included brown hydrogen, made through the ignition of coal or lignite and gray hydrogen, by reforming it from methane gas or blue hydrogen which is when gray hydrogen’s carbon emissions are captured.
What is Green Hydrogen?
Green hydrogen uses clean energy to power the electrolysis of water which separates the water molecule into hydrogen and oxygen gas. Then when the energy is ready to be used, the hydrogen is reacted with oxygen and generates energy through the movement of electrons. There are three main types of electrolyzers.
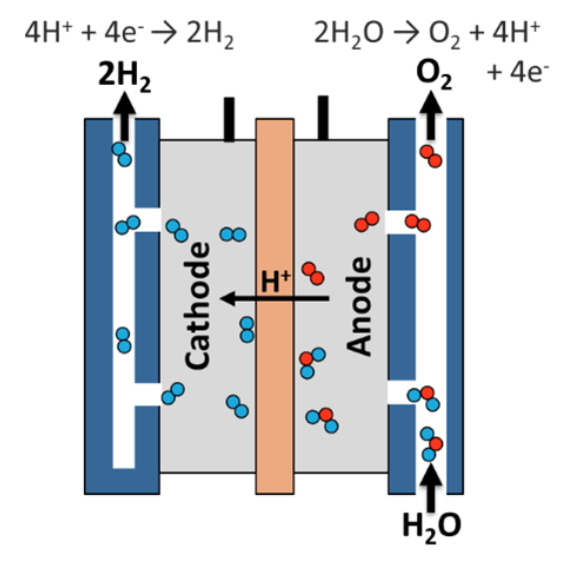
The first diagram shows a polymer electrolyte membrane electrolyzer which is the most popular because it produces high purity hydrogen and is easy to cool. In this process, water is split into protons and oxygen at the anode. Then the protons flow through the polymer electrolyte membrane and combine with electrons at the cathode to form hydrogen gas.
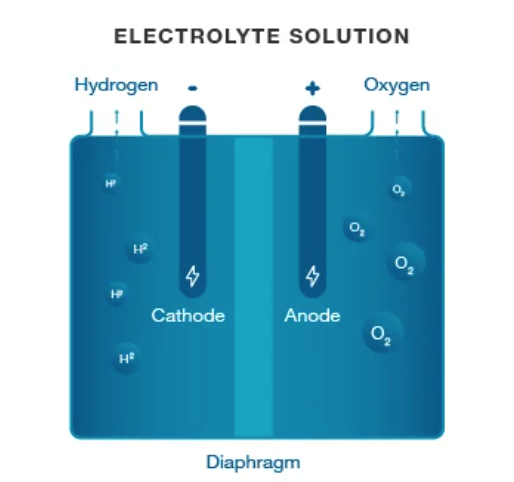
Another form of electrolyzer is an alkaline electrolyzer and this process has been in use for over 100 years. When electrical current is added to the cell, hydroxide moves from the cathode to the anode which produces hydrogen bubbles at the cathode and oxygen bubbles at the anode.
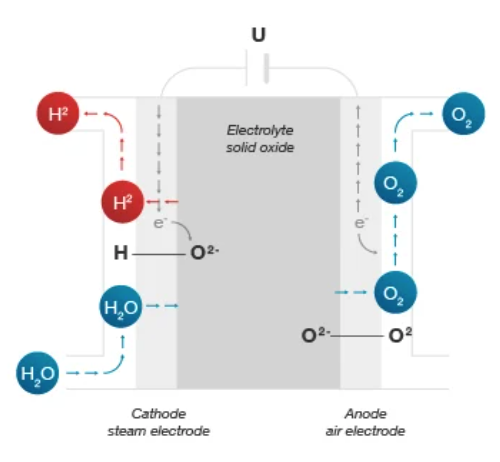
And lastly, the solid oxide electrolyzer. This electrolyzer requires a temperature between 500 and 800 degrees Celsius and can be more efficient than the other two methods in the future. The process uses a solid ceramic membrane and the electrons react with water at the cathode to form hydrogen gas and negative ions. This is the least technologically developed of the three.
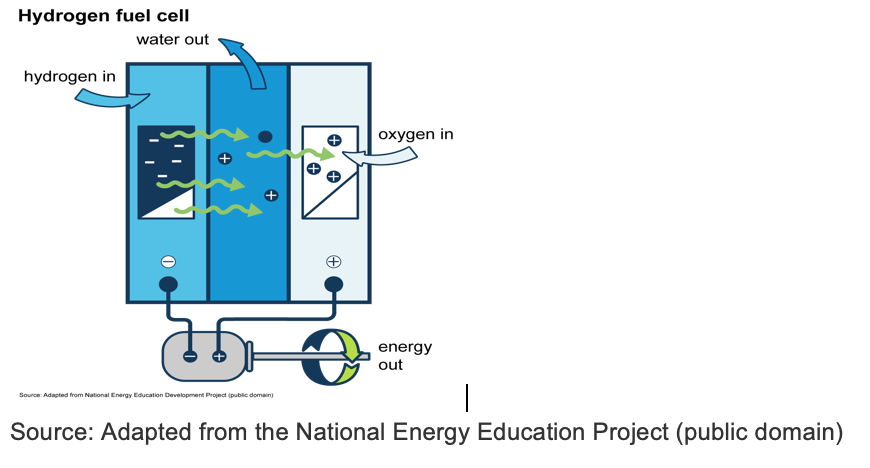
After the hydrogen gas has been produced, it can be put into a hydrogen fuel cell to store for future use. As the diagram below shows, a fuel cell consists of an anode (negative electrode), a cathode (positive electrode), and an electrolyte in between. Electrons travel from the anode to the cathode via an external circuit that produces electricity. Then the leftover protons at the anode side travel through the electrolyte to the cathode and form water.
Applications of Green Hydrogen
There are three main uses of green hydrogen as it relates to transitioning to a low carbon economy. They are energy storage, transportation, and industrial processes like steel, cement, and fertilizer production. Steel production is very carbon emission-intensive and certain companies like China BaoWu have turned to green hydrogen as a possible fuel source. As for transportation, vehicle models that run on green hydrogen are looking to enter the market. The potential for this technology is great because a hydrogen fuel cell is two to three times more efficient than an internal combustion engine that uses gasoline. Toyota is a leading company of this wave, being the first to mass produce hydrogen fuel cell cars. In the U.S., models of hydrogen fuel cell cars are also developing but due to the lack of fueling stations, they are not widespread yet.
Even more recently, green hydrogen fuel has entered the aviation industry and ZeroAvia is one of the companies in this field that will be represented at the 2022 Clean Tech Summit on March 29-30, 2022.
References
Bai, Y. J., Liu, Y. K., Wen, S. M., & Jia, D. (2021, June 21). China’s hydrogen roadmap: 4 things to know. Nikkei Asia. Retrieved November 19, 2021, from https://asia.nikkei.com/Spotlight/Caixin/China-s-hydrogen-roadmap-4-things-to-know.
Corporativa, I. (n.d.). What is an electrolyser and why is it key to green hydrogen supply? Iberdrola. Retrieved November 19, 2021, from https://www.iberdrola.com/sustainability/electrolyzer.
Deign, J. (2020, June 30). So, what exactly is green hydrogen? Greentech Media. Retrieved November 19, 2021, from https://www.greentechmedia.com/articles/read/green-hydrogen-explained.
Fuel cells. Energy.gov. (n.d.). Retrieved November 19, 2021, from https://www.energy.gov/eere/fuelcells/fuel-cells.
Hydrogen production: Electrolysis. Energy.gov. (n.d.). Retrieved November 19, 2021, from https://www.energy.gov/eere/fuelcells/hydrogen-production-electrolysis.
U.S. Energy Information Administration (EIA). (2021, January 7). Hydrogen explained. eia.gov. Retrieved November 19, 2021, from https://www.eia.gov/energyexplained/hydrogen/use-of-hydrogen.php.
About the Author
UNC Chapel Hill Class of 2024
Environmental Science Major (B.S.) with a minor in Statistics and Analytics, Dual degree with a master’s in Information Science
